Advertisements
Advertisements
Question
The overlap of orbitals involved in the formation of C - Br bond in vinyl bromide is ______.
Options
sp2 - 3pz
sp3 - 3pz
sp2 - 4pz
sp3 - 4pz
MCQ
Fill in the Blanks
Solution
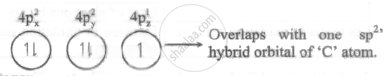
The overlap of orbitals involved in the formation of C - Br bond in vinyl bromide is sp2 - 4pz.
Explanation:
\[\begin{array}{cc}
\ce{H}\phantom{....}\ce{H}\\
|\phantom{.....}|\\
\ce{\underset{\text{vinyl bromide}}{H - C = C - Br}}\end{array}\]
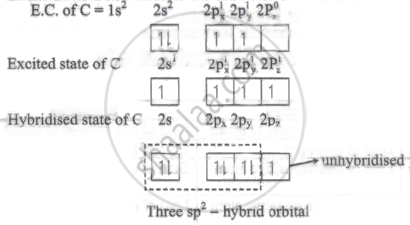
Outer E. C. of Br: [Ar] 4s2 3d10
shaalaa.com
Is there an error in this question or solution?
