Advertisements
Advertisements
Question
The process of converting a substance from solid state to gaseous state is called condensation.
Options
True
False
Solution
The process of converting a substance from a solid state to gaseous state is called condensation- False.
Correct statement:
The process of converting a substance from solid to gas is called sublimation.
APPEARS IN
RELATED QUESTIONS
Describe an experiment to show that there is absorption of heat energy when the ice melts.
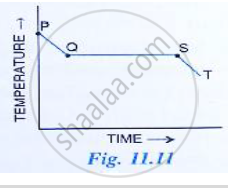
Fig 11. 11 shows the variation in temperature with time when some wax cools from the liquid phase to the solid phase.
(i) In which part of the curve, the wax is in liquid phase?
(ii) What does the part QS of the curve represent?
(iii) In which part of the curve, the wax will be the in the liquid as well as solid phase?
(iv) In which part of the curve, the wax is in solid phase?
In an experiment. 17 g of ice is used to bring down the temp. of 40 g of water from 34°C to its freezing point. The sp. heat capacity of water is 4.25 J/g°C. Calculate sp. latent heat of ice.
What mass of ice at 0°C will be required to cool 0.9 kg of water from 35°C to 0°C? Assume all the ice used melts. The specific heat capacity of water is 4.2 x 103 J/kgoC and specific latent heat of fusion of ice is 336 x 103 J/kg.
Fill in the following blank using suitable word:
1 cal = .......... J
Fill in the following blank using suitable word:
Whenever mechanical work is done, .......... Is produced.
A mass of 50 g of a certain metal at 150° C is immersed in 100 g of water at 11° C. The final temperature is 20° C. Calculate the specific heat capacity of the metal. Assume that the specific heat capacity of water is 4.2 J g-1 K-1.
Calculate the heat energy that will be released when 5.0 kg of steam at 100°C condenses to form water at 100°C. Express your answer in S.I. unit. (Specific latent heat of vaporization of steam is 2268 kj/kg.)
