Advertisements
Advertisements
Question
The total number of π-bond electrons in the following structure is ______.
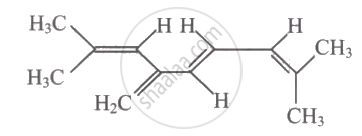
Options
4
8
12
16
MCQ
Fill in the Blanks
Solution
The total number of π-bond electrons in the following structure is 8.
Explanation:
The bonds are the C-H and C-C single bonds. The presented molecule has a C = C double bond made up of one π bond and one π-bond. Eight σ-bond electrons are shared between four π bonds.
shaalaa.com
Alkenes - Structure of Double Bond (Ethene)
Is there an error in this question or solution?
