Advertisements
Advertisements
Question
Using the Ellingham diagram,
(A) Predict the conditions under which
- Aluminium might be expected to reduce magnesia.
- Magnesium could reduce alumina.
(B) it is possible to reduce Fe2O3 by coke at a temperature around 1200 K
Answer in Brief
Solution
(A)
i. Ellingham diagram for the formation of Al2O3 and MgO intersects around 1600 K. Above this temperature, aluminium line lies below the magnesium line. Hence we can use aluminium to reduce magnesia above 1600 K.
ii. In the Ellingham diagram below 1600 K magnesium line lies below the aluminium line. Hence below 1600 K magnesium can reduce alumina.
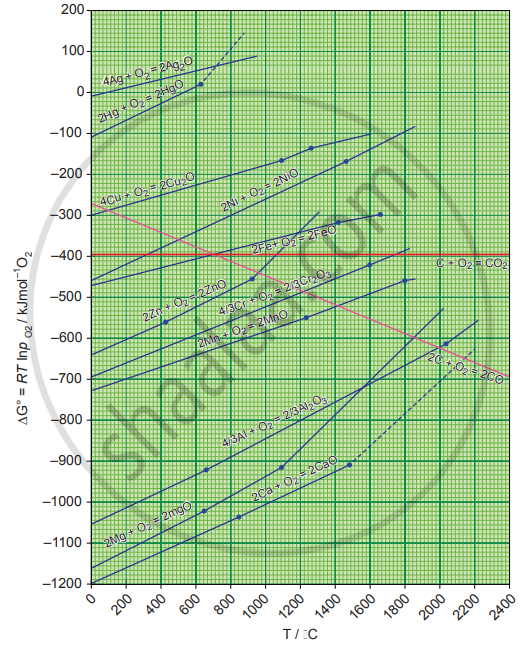
(B) In the Ellingham diagram above 1000 K carbon line lies below the iron line. Hence it is possible to reduce Fe2O3 by coke at a temperature around 1200 K.
shaalaa.com
Is there an error in this question or solution?
