Advertisements
Advertisements
Questions
Very short answer question.
What is osmotic pressure?
Explain the term osmosis.
Solution
i. The pressure exerted due to osmosis is called osmotic pressure.
ii. Osmotic pressure is a pressure of the solution, which is required in opposite direction, so as to stop the entry of solvent molecules into the cell.
OR
Osmotic pressure of a solution is equivalent to the pressure which must be exerted upon it to prevent flow of solvent across a semipermeable membrane.
RELATED QUESTIONS
Define the following:
Turgor pressure
The diagram given below represents an experimental set – up to demonstrate a certain process. Study the same and answer the questions that follow:
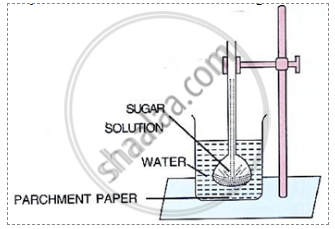
what would you observe in the experimental set-up after an hour or so?
The diagram given below represents an experimental set – up to demonstrate a certain process. Study the same and answer the questions that follow:

Mention two advantages of the process to the plants.
Fill in the Blank
Water and mineral salts absorbed by root is known as ______.
Osmosis is a property of ______.
Answer the following question.
Define and or explain the term:
Diffusion
Answer the following question.
Define and or explain the term:
Wall pressure
Answer the following question.
Distinguish between Osmotic pressure and Turgor pressure.
Cell wall is _______.
Aquaporins (water channels) are involved in ______ type of transport.
