Advertisements
Advertisements
Question
Visha took 4 test-tubes namely A, B, C & D containing \[\ce{CH3CH = CH2}\], \[\ce{CH3CH2CH = CH2}\], \[\ce{CH3CH = CH - CH3}\] and \[\ce{(CH3)2C = CH2}\] respectively and tried to convert them into tertbutylalcohol. She carried out acid catalysed hydration reaction on every alkene. Out of the four test-tubes, the one which will give desired result is:
Options
A
B
C
D
MCQ
Solution
D
Explanation:
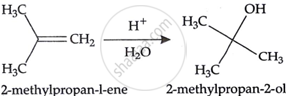
shaalaa.com
Is there an error in this question or solution?
