Advertisements
Advertisements
Question
What is the difference between the order of a reaction and the molecularity of a reaction?
Solution
| order of a reaction | molecularity of a reaction |
| Order of a reaction is an experimental quantity. It can be zero and even a fraction | Molecularity of a reaction is the number of species (atoms, ions or molecules) taking part in the elementary reaction. It cannot be zero or non-integer |
| The order is applicable to elementary as well as complex reactions. | Molecularity is applicable only for elementary reactions |
| For complex reaction, the order is given by slowest step. |
Molecularity of the slowest step is equal to the order of overall reaction |
APPEARS IN
RELATED QUESTIONS
Fill in the blanks by choosing the appropriate word/words from those given in the brackets:
When the concentration of a reactant of the first-order reaction is doubled, the rate becomes ……… times, but for ……….. order reaction, the rate remains the same.
(Henry’s, aldol condensation, absence, do not, ohm, Raoult’s, increases, common ion effect, easily, three, solubility product, ohm-1, two, four, ohm-1, cm2, Cannizzaro, ohm-1 cm-1, zero, decreases, presence)
What is the order of reaction whose rate constant has the same unit as the rate of reaction?
Answer the following question:
Identify the order of reaction from each of the following units of rate constant (k):
(i) mol L-1 sec-1
(ii) mol-1 L sec-1
Answer the following question:
Name the order of reaction which proceeds with a uniform rate throughout.
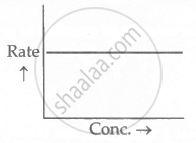
For a chemical reaction, the rate with concentration is shown. What is the order of reaction?