Advertisements
Advertisements
Question
What do you mean by the inter-molecular spaces? How do they vary in different states of matter?
Solution
INTER-MOLECULAR SPACES “The spacing between the molecules of the matter is called Inter-molecular spaces.”
The inter-molecular spaces in different states of matter:
- The intermolecular space between solids is absent.
- The intermolecular space between Liquid is moderate but present.
- The intermolecular space between gasses is free-flowing and plenty.
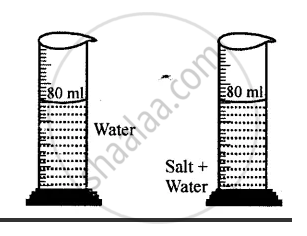
Explanation of inter-molecular space: Take water in a measuring cylinder say up to 80 ml. mark. Add 10 gm of salt to it. The volume in the cylinder should increase. On dissolving salt we find volume remains same i.e. up to 80 ml mark. This is because there are spaces in water molecules and salt molecules occupy these spaces and volume remains the same.
APPEARS IN
RELATED QUESTIONS
Name a metal which has no fixed shape.
Which of the following are not compounds ?
Chlorine gas, Potassium chloride, Iron powder, Iron sulphide, Aluminium foil, Iodine vapour, Graphite, Carbon monoxide, Sulphur powder, Diamond
Which of the following are compounds ?
- CO
- No
- NO
- Co
On the basis of composition of matter, milk is considered to be :
In the following set of substances, one item does not belong to the set. Select this item and explain why it does not belong to the set :
Hydrogen, Oxygen, Steam, Chlorine
There are three substances X, Y and Z. The substance X does not have a fixed melting point or boiling point and it still shows the individual properties of its constituents. The substance Y is a pure substance which occurs in nature as such. The substance Y has a fixed melting point and boiling point but it cannot be broken down into simpler substances by any chemical means. The substance Z is also a pure substance whose properties are entirely different from those of its constituents. The substance Z can, however, be divided by electrolysis into two substances which belong to the same class of substances as Y.
- What type of substance could X be ? Name one substance like X.
- What type of substance could Y be ? Name one substance like Y.
- What type of substance could Z be ? Name one substance like Z.
- Which process involves absorption or release of an appreciable amount of energy : formation of substance X or formation of substance Z ?
- Name the three groups into which all the substances like Y are divided on the basis of their properties.
A, B and C are all liquids. Liquid A has a comparatively low boiling point. On heating, liquid A vaporises completely without leaving behind any residue. Liquid A is being used increasingly as a fuel in motor vehicles either alone or by mixing with petrol. Liquid B has a very high boiling point. It also vaporises completely on heating, without leaving any residue. Liquid B is a conductor of electricity and used in making thermometers. Liquid C has a moderate boiling point. On heating, liquid C vaporises leaving behind a white solid D which is used in cooking vegetables. The condensation of vapours from C give a liquid E which turns anhydrous CuSO4 to blue.
(a) Which liquid could be an element ? Name this element.
(b) Which liquid could be a mixture ? Name this mixture.
(c) Which liquid could be a compound ? Name this compound.
(d) What could the solid D be ?
(e) What do you think is liquid E ?
What is the concentration of a solution which contains 16 g of urea in 120 g of solution ?
Fill in the following blanks with suitable words :
A heterogeneous mixture of liquid and solid is conveniently separated by...............
Define the term matter.
