Advertisements
Advertisements
Question
What do you understand by Temporary hardness?
Solution
Temporary hardness is water hardness due to the presence of calcium and magnesium carbonates and bicarbonates, which can be precipitated by heating the water. It can be removed by processes such as boiling or lime softening, and then separation of water from the resulting precipitate.
Temporary hardness is very common and is responsible for the deposition of calcium carbonate scale in pipes and equipment. These deposit formations lead to clogged plumbing and reduced efficiency of heat exchangers.
Temporary hardness is also known as carbonate hardness.
APPEARS IN
RELATED QUESTIONS
Give reason On opening a bottle of a cold drink, a fizz sound is heard.
What are its causes and how can water pollution be controlled ?
Name the solute, solvent & solution in the statement – ‘sodium chloride dissolves in water to give sodium chloride solution’. Define each of the terms in italics.
What is meant by the term ‘offshore drilling’. State the main environmental effects of offshore drilling.
If, at some place, it did not rain for a very long period of time, what would be its effect on the life of the people there?
True or false? Correct the wrong statement.
We get rainwater throughout the year.
“Every Drop Counts” is a slogan related to
From where do the following usually get water? In which form is water present in them?
Aquifer
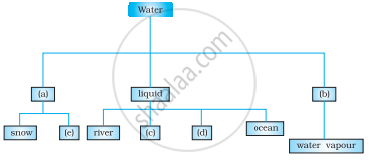
Complete the following chart by writing appropriate words in the boxes marked (a) to (e).
A man travelling on a train threw an empty packet of food on the platform. Do you think this is a proper waste disposal method? Elaborate.
