Advertisements
Advertisements
Question
What is the highest oxidation state possessed by phosphorus in it's oxyacids?
Options
+5
+3
+6
+4
MCQ
Solution
+5
Explanation:
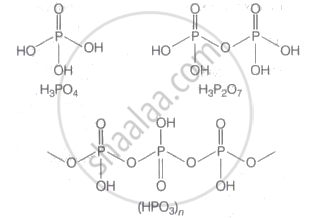
+ 5 is the highest oxidation state possessed by phosphorus in it's oxyacids. In oxoacids phosphorus is tetrahedrally surrounded by other atoms. All these acid contains at least one P = O bond and one P-OH bond. The oxoacids in which phosphorus has maximum oxidation state (+ 5) is orthophosphoric (H3PO4), pyrophosphoric (H4P2O7) and metaphosphoric (HPO3)n·
shaalaa.com
Chemical Properties of Elements of Groups 16, 17 and 18
Is there an error in this question or solution?
