Advertisements
Advertisements
Question
What is the molar mass of a compound represented below?
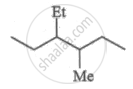
Options
108 g mol-1
120 g mol-1
128 g mol-1
126 g mol-1
MCQ
Solution
128 g mol-1
Explanation:

M.F. = C9H20
No. of 'C' atoms = 9
No. of 'H' atoms = 20
Molecular mass = (9 × 12) + (1 × 20) = 128 g mol-1
shaalaa.com
Structural Representation of Organic Molecules
Is there an error in this question or solution?
