Advertisements
Advertisements
Question
What is the shape and magnetic nature of permanganate ion?
Options
Pyramidal diamagnetic
Tetrahedral, diamagnetic
Tetrahedral, paramagnetic
Planar, paramagnetic
MCQ
Solution
Tetrahedral, diamagnetic
Explanation:
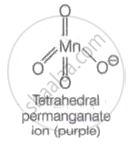
The ions of permanganate are tetrahedral. The π- bonding occurs when the p-orbitals of oxygen collide with the d-orbitals of manganese. Because there are no unpaired electrons, it is also diamagnetic. The following is the structure of the permanganate ion:
shaalaa.com
Magnetic Properties of Solids
Is there an error in this question or solution?
