Advertisements
Advertisements
Question
What is the type of magnetic behavior and geometry respectively in Cuproammonium sulphate (Atomic number of Cu = 29)?
Options
Paramagnetic and square planar
Paramagnetic and tetrahedral
Diamagnetic and pyramidal
Diamagnetic and tetrahedral
MCQ
Solution
Paramagnetic and square planar
Explanation:
Cuproammonium sulphate - \[\ce{[Cu(NH3)4]SO4 . H2O}\]
[Cu(NH3)4]2+ → Oxidation state of Cu = +2
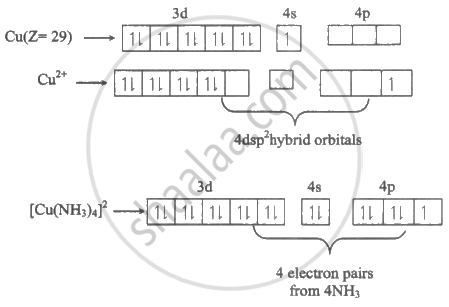
dsp2 - Square planar, Paramagnetic - One unpaired electron.
shaalaa.com
Theories of Bonding in Complexes
Is there an error in this question or solution?
