Advertisements
Advertisements
Question
What will be the work done, when one gram of ice at -10° C is converted into steam at 100°?
Options
3045 J
6056 J
721 J
616 J
MCQ
Solution
3045 J
Explanation:
Ice (- 10° C) converts into steam as follows (ci = Specific heat of ice, cw= Specific heat of water, Lf = Latent heat of fusion and Lv = Latent beat of vaporization)
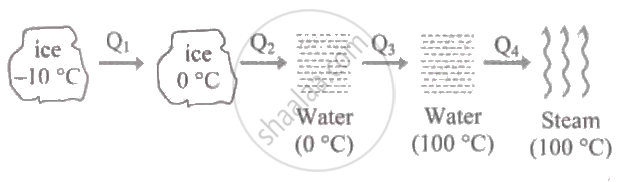
Total heat required
`"Q" = "Q"_1 + "Q"_2 + "Q"_3 + "Q"_4`
`= "mc"_"i" Delta theta_1 + "mL"_"f" + "mc"_"W" Delta theta_2 + "mL"_"V"`
`= 1 xx 0.5 (10) + 1 xx 80 + 1 xx 1 xx (10 - 0) + 1 xx 540`
`= 725 "cal"`
Work done W = JQ
= 4.2 x 725
= 3045 J
shaalaa.com
Change of State
Is there an error in this question or solution?
