Advertisements
Advertisements
Question
When a gas jar containing colourless air is kept upside down over a gas jar full of brown-coloured bromine vapour, then after some time, the brown colour of bromine vapour spreads into the upper gas jar making both the gas jars appear brown in colour. Which of the following conclusion obtained from these observations is incorrect?
Options
bromine vapour is made of tiny particles which are moving
air is made up of tiny particles which are moving
the particles of bromine are moving but those of air are not moving
even though bromine vapour is heavier that air, it can move up against gravity
Solution
The particles of bromine are moving but those of air are not moving
The statement that the particles of bromine are moving but those of air are not moving is incorrect because the particles of matter are constantly in motion. It appears as if the air molecules are not moving because air is colourless.
APPEARS IN
RELATED QUESTIONS
Give reasons:
We can easily move our hand in air, but to do the same through a solid block of wood, we need a karate expert.
Give a reason for the following observation.
We can get the smell of perfume sitting several metres away.
Name the physical state of matter which can be easily compressed.
Write the full forms of the following
- LPG
- CNG
A bottle of perfume was opened in a room. The smell of its vapours spread in the entire room. Name the property of gases which is responsible for this behaviour of perfume vapours.
Fill in the following blank with suitable words :
Solid, liquid and gas are the three ........................ of matter.
When an incense stick (agarbatti) is lighted in one corner of a room, its fragrance quickly spreads in the entire room. Name the process involved in this.
The space between ______ particles is the greatest one.
 |
|
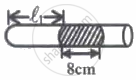
| Fig. I | Fig. II |
In figure-I, an air column of length ℓ1 is entrapped by a column of Hg of length 8 cm. In figure-II length of same air column at the same temperature is ℓ2. The `ℓ_1/ℓ_2` is:
(1 atm = 76 cm of Hg)
A gaseous mixture of He and O2 is found to have a density of 0.518 gL-1 at 25° C and 720 torr. The mass percent of helium in this mixture is ______.