Advertisements
Advertisements
Question
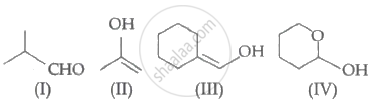
Which among the above compound/s does/do not form Silver mirror when treated with Tollen's reagent?
Options
Only (II)
(I), (III) and (IV) only
(III) and (IV) only
Only (IV)
Solution
Only (II)
Explanation:
Aldehydes are identified using Tollen's reagent. An aldehyde is converted into the equivalent carboxylic acid. This is due to carbonyl C's attachment to an H atom in an aldehyde. Hence, aldehydes are readily oxidised by Tollen's reagent-producing silver mirror. The ketones do not contain any H atom directly attached to carbonyl C making it inert for this oxidation. Only α-hydroxy ketone will give a positive silver mirror test. Hemiacetals also give positive silver mirror tests.
Compound I is an aldehyde, hence it will give a positive silver mirror test.
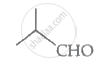
Compound II tautomerises to give a ketone, which is not an α - hydroxy ketone. Thus, compound II will give a negative silver mirror test.
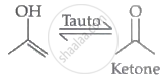
Compound III tautomerises to give an aldehyde. Therefore, compound Ill gives a positive silver mirror test.

Compound IV is an hemiacetal thus, gives a positive silver mirror test.
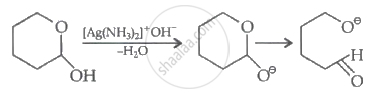
Therefore, compound II will not form silver mirror when treated with Tollen's reagent
