Advertisements
Advertisements
Question
Which among the following pairs of halogen forms the interhalogen compound of the type \[\ce{XX^{'}_7}\]?
Options
Cl and F
I and F
I and Cl
Br and F
MCQ
Solution
I and F
Explanation:
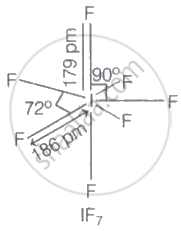
The interhalogen compound of type \[\ce{XX^{'}_7}\], iodine heptafluoride with the chemical formula IF7, is made up of the halogen pairs iodine (I) and fluorine (F).
VSEPR theory predicts that it has an unusual pentagonal bipyramidal structure.
shaalaa.com
Is there an error in this question or solution?
