Advertisements
Advertisements
Question
Which among the following phenolic compound is most acidic in nature?
Options
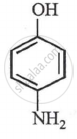
p-aminophenol
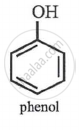
Phenol
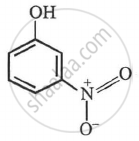
m-nitrophenol
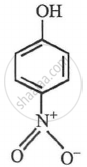
p-nitrophenol
MCQ
Solution
p-nitrophenol
Explanation:
The order for acidity in phenols depends upon the position of electron withdrawing groups. \[\ce{- NH2}\] is an electron releasing group. Hence, it decreases the acidity of phenol.
p-nitrophenol > m-nitrophenol > phenol > p-aminophenol.
 |
 |
 |
 |
| (-I-effect) (-R-effect) para nitro phenol |
(Only -I-effect) meta nitro phenol |
(+I-effect) (+R-effect) p-amino phenol |
shaalaa.com
Alcohols and Phenols
Is there an error in this question or solution?
