Advertisements
Advertisements
Question
Which is low spin complex?
Options
\[\ce{Fe(CN)^{4-}_6}\]
\[\ce{Co(NO2)^{3-}_6}\]
\[\ce{Mn(CN)^{3-}_6}\]
All of these
MCQ
Fill in the Blanks
Solution
All of these.
Explanation:
| Fe(26) | 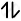 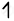 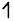 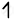 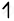 |
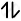 |
| 3d | 4s | |
| Fe+2 | 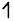 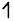 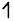 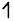 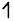 |
CN- is strong field ligand, it pair the electron and form a low spin complex.
| Co(27) | 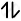 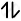 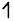 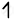 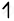 |
|
| 3d | 4s | |
| Co+3 | 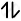 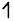 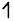 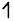 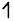 |
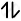 |
| 3d | 4s |
`"NO"_2^-` is able to cause the pair pairing of electrons, so a low spin complex will form.
| Mn(25) | 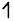 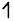 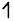 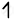 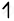 |
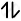 |
| 3d | 4s | |
| Mn+3 | 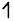 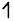 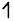 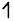 |
|
| 3d | 4s |
CN- strong field ligand, causes the pairing of electron.
shaalaa.com
Is there an error in this question or solution?
