Advertisements
Advertisements
Question
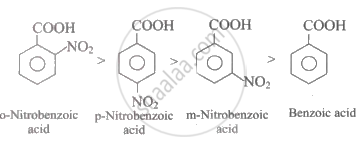
Which of the following carboxylic acids will have the highest acidity?
Options
o-Nitrobenzoic acid
p-Nitrobenzoic acid
m-Nitrobenzoic acid
Benzoic acid
Solution
o-Nitrobenzoic acid
Explanation:
An aromatic carboxylic acid, electron-withdrawing groups like −NO2 increases the acidity of the acid. Nitro group has a very strong electron-withdrawing resonance effect (−R effect) as well as electron withdrawing inductive effect (−I effect). However, due to ortho-effect, o-nitrobenzoic acid is the strongest acid amongst all the given acids. In p-nitrobenzoic acid, both −R and −I effect of the −NO2 group increases the acidity while in m-nitrobenzoic acid only the weaker −I effect increases the acidity. Thus, the decreasing order for acidity in aromatic carboxylic acids is given by: