Advertisements
Advertisements
Question
Which of the following is a weaker base than ammonia?
Options
\[\ce{CH3-NH2}\]
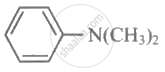
\[\ce{(CH3CH2)3N}\]
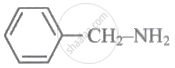
MCQ
Solution

Explanation:
Arylamines are weaker bases than ammonia.
shaalaa.com
Basicity of Amines
Is there an error in this question or solution?
