Advertisements
Advertisements
Question
Which of the following is an aromatic compound?
Options
MCQ
Solution
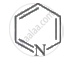
Explanation:
(A)
Because it possesses 4π electrons, the compound is non-aromatic and so does not follow the Huckel rule of aromaticity.
(B)

Because it has 4π electrons, the compound is non-aromatic and so does not follow the Huckel rule of aromaticity.
(C)

Because it has 6π electrons, the compound is aromatic and follows the Huckel rule of aromaticity.
(D)

Even though it follows Huckel's aromaticity rule, the compound is non-aromatic. This is due to the fact that one of the carbon atoms is sp3 hybridised and lacks a p orbital, making complete overlap around the ring impossible.
shaalaa.com
Aromatic Hydrocarbons
Is there an error in this question or solution?
