Advertisements
Advertisements
Question
Which of the following is NOT an example of redox reaction?
Options
\[\ce{HF + OH^- -> H2O + F^-}\]
\[\ce{3H3AsO3 + BrO^-_3 -> Br^- + 3H3AsO4}\]
\[\ce{I2 + 2S2O^{2-}_3 -> S4O^{2-}_6 + 2I^-}\]
\[\ce{C + O2 -> CO2}\]
MCQ
Solution
\[\ce{HF + OH^- -> H2O + F^-}\]
Explanation:
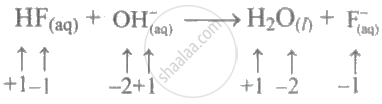
In the above process, no species oxidation number changes. As a result, this isn't a redox reaction in the traditional sense.
shaalaa.com
Oxidation Number
Is there an error in this question or solution?
