Advertisements
Advertisements
Question
Which of the following molecular formula represents Marshall's acid?
Options
H2S2O7
H2S2O6
H2S2O8
H2SO5
MCQ
Solution
H2S2O8
Explanation:
Peroxodisulphuric acid (H2S2O8) represents Marshall's acid. It is a inorganic compound. It contain sulphur in its + 6oxidation state and a peroxide group. It's structure is as follows:
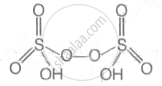
Peroxodisulphuric acid (H2S2O8)
shaalaa.com
Oxoacids
Is there an error in this question or solution?
