Advertisements
Advertisements
Question
Which of the following species can act as the strongest base?
Options
\[\ce{OH-}\]
\[\ce{C6H5O-}\]
\[\ce{RO-}\]
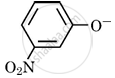
MCQ
Solution
\[\ce{RO-}\]
Explanation:
The stronger base is the conjugate form of the weak acid. \[\ce{ROH}\] is the weakest acid. The alkyl group in \[\ce{RO-}\] has a positive inductive action, increasing the negative charge and making it a strong base.
shaalaa.com
Is there an error in this question or solution?
