Advertisements
Advertisements
Question
Which of the following statement is incorrect?
Options
\[\ce{[NiCl4]^{2-}}\] is paramagnetic.
\[\ce{[Ni(CO)4]}\] is diamagnetic.
\[\ce{[Cr(NH3)6]^{3+}}\] is paramagnetic.
\[\ce{[Ni(CN)4]^{2-}}\] is paramagnetic.
MCQ
Solution
\[\ce{[Ni(CN)4]^{2-}}\] is paramagnetic.
Explanation:
\[\ce{[Ni(CN)4]^{2-}}\] is diamagnetic not paramagnetic.
Ni atom (Z = 28) (ground state) = \[\ce{[Ar] 3d^8, 4s^2, 4p^0}\]
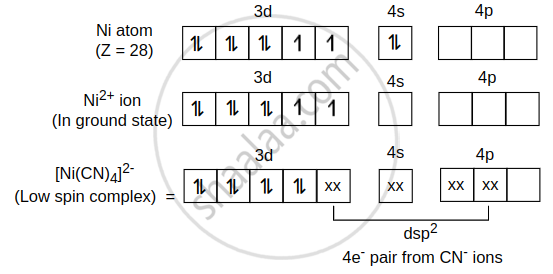
CN- is a strong field ligand, due to which pairing of electron takes place.
shaalaa.com
Magnetic Properties of Solids
Is there an error in this question or solution?
