Advertisements
Advertisements
Question
Which one of the following is the correct set with respect to molecule, hybridization and shape?
Options
BeCl2, sp2, linear
BeCl2, sp2, triangular planar
BCl3, sp2, triangular planar
BCl3, sp3, tetrahedral
MCQ
Solution
BCl3, sp2, triangular planar
Explanation:
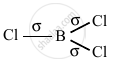
no lone pair, sp2 hybridization, triangular planar
shaalaa.com
Is there an error in this question or solution?
