Advertisements
Advertisements
Question
Which one of the following reactions indicated the reducing ability of hydrogen peroxide in basic medium?
Options
\[\ce{HOCl + H2O2 -> H2O^+ + Cl^- + O2}\]
\[\ce{PbS + 4H2O2 -> PbSO4 + 4H2O}\]
\[\ce{2MnO^-4 + 2H2O2 -> 2MnO2 + 3O2 + 2H2O + 2OH^-}\]
\[\ce{Mn^{2+} + H2O2 -> Mn^{4+} + 2OH^-}\]
MCQ
Solution
\[\ce{2MnO^-4 + 2H2O2 -> 2MnO2 + 3O2 + 2H2O + 2OH^-}\]
Explanation:
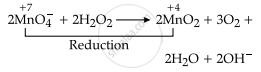
Since the oxidation state of Mn changes from +7 to +4. So, Mn gets reduced.
Therefore, H2O2 is acting as a reducing agent in the above reaction.
shaalaa.com
Chemical Properties of Hydrogen Peroxide
Is there an error in this question or solution?
