Advertisements
Advertisements
Question
Which one of the following substituents at para-position is most effective in stabilizing the phenoxide 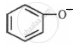 ion?
ion?
Options
−CH3
−OCH3
−COCH3
−CH2OH
MCQ
Solution
−COCH3
Explanation:
Because of charge delocalisation, the electron-withdrawing group stabilises the benzene ring.
−CH3 and −CH2OH are electron-donating groups that reduce benzene ring stability. −OCH3 is a weaker electron withdrawing group than −COCH3.
As a result, the −COCH3 group helps to stabilise the phenoxide ion at the para-position.
shaalaa.com
Chemical Properties of Aldehydes and Ketones
Is there an error in this question or solution?
