Advertisements
Advertisements
Question
Which pair of species having identical shapes?
Options
BeCl2, XeF2, CO2
PF5, IF5, IF7
BF3, NH3, PCl3
CF4, SF4, XeF4
MCQ
Solution
BeCl2, XeF2, CO2
Explanation:
| Type of hybridization (H) | Geometry | |
| BeCl2 | \[\ce{Cl\overset{s}{-} Be \overset{p}{-}Cl}\] | Linear |
| H = `1/2` (2 + 2 − 0 + 0) = 2 | ||
| H = 2 = sp | ||
| XeF2 | 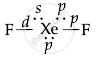 |
Linear |
| H = `1/2` (8 + 2 − 0 + 0) = 5 | ||
| H = 5 = sp3d | ||
| CO2 | \[\ce{O \overset{s}{=} C \overset{p}{=} O}\] | Linear |
| H = `1/2` ()4 + 0 − 0 + 0 = 2 | ||
| H = 2 = sp |
shaalaa.com
Is there an error in this question or solution?
