Advertisements
Advertisements
Question
Why are organic compounds studies as a separate branch of chemistry?
Solution 1
Due to the unique nature of carbon atom, it gives rise to formation of large number of compounds. Thus this demands a separate branch of chemistry.
Solution 2
Organic compounds, like inorganic compounds, follow the same fundamental chemistry laws. However, organic compounds have unique characteristics such as high number, tetravalency of carbon, composition, linkage, melting and boiling points, stability, combustibility, origin, isomerism, and nature of reactions, necessitating separate study.
Notes
Students can refer to the provided solutions based on their preferred marks.
APPEARS IN
RELATED QUESTIONS
The equation for the reaction when compound A is bubbled through bromine dissolved in carbon tetrachloride is as follows:
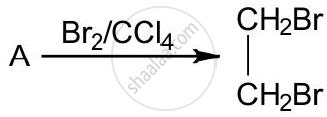
1) Draw the structure of A.
2) State your observation during this reaction.
Give the structural formula of the alcohols.
Give the reactions of ethyne with Hydrogen
Explain the following:
Ethane does not undergo addition reactions
When ethanol is heated with conc. H2SO4 at 170oC, it gets converted into ethane. In this reaction conc. H2SO4 acts as
(a) Draw the structural formula of ethene.
(b) What is the feature of the ethene structure, which allows ethene to react with chlorine in the way it does?
Draw the structural formula of ethyne.
Name the organic compound prepared by each of the following reactions:
\[\ce{CH3I + 2H- ->}\]
Compound A is bubbled through bromine dissolved in carbon tetrachloride and the product is CH2Br - CH2Br.
\[\ce{A ->[Br2/Od4] CH2Br - CH2Br}\]
Name (not formula) the compound formed when steam reacts with A in the presence of phosphoric acid.
Identify the gas evolved when sodium propionate is heated with sodalime
