Advertisements
Advertisements
Question
Write a balanced chemical equation for each of the following reactions :
Chlorine gas is treated with ethane.
Solution
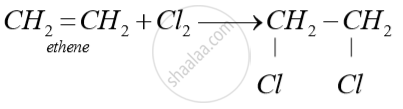
APPEARS IN
RELATED QUESTIONS
Classify the C3H6 compound as alkanes, alkenes, and alkynes.
Under what conditions does ethane get converted to acetaldehyde?
Write the electron dot formula of ethane.
The molecules of the alkene family are represented by a general formula CnH2n. Answer the following:
What is the molecular formula of alkene when n = 4?
The molecules of the alkene family are represented by a general formula CnH2n. Answer the following:
What is the structural formula of the third member of the alkene family?
How is ethene prepared by a dehydrohalogenation reaction? Give an equation and name the products formed.
Write the name and formula of the product formed in the case below:
\[\ce{H2C = CH2 + HBr}\] →
How is the structure of alkynes different from that of alkenes?
Fill in blanks with correct words from the brackets:
Alkenes are the__________(Analogous/homologous) sries of _________(saturated/unsaturated) hydrocarbons. They differ from alkanes due to the presence of_______(double/single) bonds. Alkenes mainly undergo________(addition/substitution) reactions.
Write a balanced equation:
Preparation of ethylene from ethyl alcohol.
