Advertisements
Advertisements
Question
Write a brief note on conducting polymers.
Solution
Polymers are insulated because of the absence of free electrons. In becoming electrically conductive, a polymer has to imitate metal that is the electron needs to be free to move. Such type of polymer are called Conducting polymer. Polymers with conjugate rr-electron backbones display unusual electronic properties such as low energy optical transition, low ionization potentials and high electron affinities. The result is a class of polymers that can be oxidised or reduced more easily and more reversibly than conventional polymers. The effect of this oxidation or reduction on polymer is called doping i.e., convert an insulating polymer to conducting one.
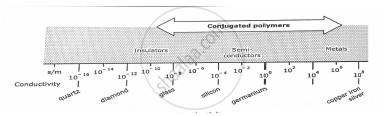
Two conditions for a polvmer to become conducting are:-
1. Polymer possess conjugate double bonds.
2. Polymer has to be distributed either by removing or adding electron to the material. This
process is called doping.
