Advertisements
Advertisements
Question
Write preparation ,properties and uses of following polymers : i) Kevlar ii) Silicone rubber iii) Buna S
Solution
(i)Kevlar
Preparation :
It is aromatic polyamide or polyaramide resin .It is prepared by polycondensation reaction between aromatic dichloride and aromatic diamines .

Properties :
1) Exceptionally strong ( 5 times stronger than steel ) .
2) High heat stability and flexibility .
3) Very high rigidity ( due to delocalized bonding which causes benzene rings to be inflexible ) .
4) High electron density in chains of Kevlar .
Uses :
1) Aerospace and Aircraft industry .
2) Making ropes ,cables ,protective clothings ,bullet proof vests,etc
3) Making motorcycle helmets .
4) Car parts . eg brakes ,tyres ,clutch linings ,etc .
(ii) Silicone rubber
Preparation : It is a type of inorganic polymer where backbone contains atoms other than carbon ,linked together by covalent bonds (-Si-O-Si-). Silicon rubbers are produced by polymerization of dimethylsilicon hydroxide .
Properties :
1) Exceptional resistance to prolonged exposure to sunlight ,weathering ,moist oils ,dilute acids and alkalis .
2) It shows flexibility in temperature from 90-250 C
3) When silicone rubber is kept at very high temperatures , it decomposes leaving behind non-conducting silica ( SiO2 ) .
4) Non-toxic in nature .
5) Water repellent .
6) Shows good electrical insulating properties .

Uses :
1) In fighter aircrafts .
2) For manufacture of tyres .
3) As adhesives in electronic industry .
4) For making artificial heart valves , padding in plastic surgery .
5) High voltage insulators .
(iii)Buna S : Preparation :Buna S / Styrene Butadiene rubber is synthesized from two monomers , namely i) Styrene (25% by weight ) ii) Butadiene ( 75% by weight ) .It is obtained by co-polymerization reaction in presence of Na as catalyst .
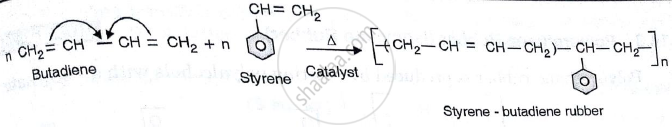
Properties :
1) High abrasion resistance .
2) High load bearing capacity and resilience .
3) Readily it gets oxidized in presence of ozone .
4) It swells in oils and solvents .
5) It needs more accelerators for vulcanization .
6) It can be vulcanized by sulphur but the quantity required is less .
Uses :
1) It is used in motorcycle tyres .
2) It is used in shoe soles , foot wear components , floor tiles.
3) Wire and cable insulations , adhesives .
