Advertisements
Advertisements
Question
Write the molecular formulae of compound calcium oxide.
Solution
Compound Calcium oxide is formed of elements calcium (Ca) and oxygen (O)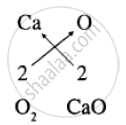
Symbols combining power Here the subscript number is same Ca2 Formula of calcium oxide is CaO
APPEARS IN
RELATED QUESTIONS
Name the process by which the components of following mixtures can be separated.
- Iron and sulphur
- Ammonium chloride and sand
- Common salt from sea water
- Chaff and grain
- Water and mustard oil
- Sugar and water
What are the different types of mixtures ?
The methods of separating components of a given mixture are based on the
What is the proportion of element present in the following compound?
CO2
Name two compounds which dissolve in water.
State the information obtained from the formula of a compound.
Explain the term compounds.
Tabulate a comparative chart – to differentiate between elements, compounds, and mixtures. Differentiate them with reference to existence
Tabulate a comparative chart – to differentiate between elements, compounds, and mixtures. Differentiate them with reference to properties
What is the molecular formula of the following compound?
glucose
Explain the term mixture.
Match the following.
| Iron | For making wires |
| Copper | Sewing needle |
| Tungsten | As a fuel for ignition in rocket |
| Boron | Making the filament of a bulb |
State whether true or false. If false, correct the statement.
Aspirin is composed of 60% Carbon, 4.5% Hydrogen and 35.5% Oxygen by mass. Aspirin is a mixture.
Non-metals are usually poor conductors of heat and electricity. They are non-lustrous, non-sonorous, non-malleable and are coloured.
Name a non-metal which is known to form the largest number of compounds.
Classify the following as element and compound,
- Silver
- Ammonia
- Water
- Mercury.
Complete the table
| Compound | Constituent elements |
| Sodium carbonate | Sodium, Carbon. Oxygen |
| Water | |
| Calcium oxide | |
| Sugar | Carbon, Hydrogen, Oxygen |
When does the pressure of the gas increase?
A compound has a definite ______.
