Advertisements
Advertisements
प्रश्न
A bottle of perfume was opened in a room. The smell of its vapours spread in the entire room. Name the property of gases which is responsible for this behaviour of perfume vapours.
उत्तर
When a bottle of perfume in opened, fragrance of perfume spreads in the whole room. This behaviour is due to larger spaces in between the tiny particles of gas and their random movements which leads to the process of diffusion.
APPEARS IN
संबंधित प्रश्न
Give a reason for the following observation.
We can get the smell of perfume sitting several metres away.
Why do gases have neither a fixed shape nor a fixed volume?
A girl is cooking some food in the kitchen. The smell of food being cooked soon reaches her brother's room. Explain how the smell could have reached her brother's room.
Give one example of diffusion of gases in a liquid.
Explain how, the smell of food being cooked in the kitchen reaches us even from a considerable distance.
Explain why, diffusion occurs more quickly in a gas than in a liquid.
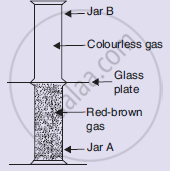
Look at the diagram on the right side. Jar A contains a red-brown gas whereas jar B contains a colourless gas. The two gas jars are separated by a galas plate placed between them
- What will happen when the glass plate between the two jars is pulled away?
- What name is given to the phenomenon which takes place?
- Name the brown gas which could be in jar A.
- Which is the colourless gas most likely to be present in jar B?
- Name one coloured solid and one colourless liquid which can show the same phenomenon.
Why do gases not have fixed shape?
Comment upon the following:-
shape
Which of the following is TRUE about gaseous state?
