Advertisements
Advertisements
प्रश्न
Why do gases not have fixed shape?
उत्तर
- Gases possess more intermolecular space and less forces of attraction.
- They can move freely at any direction.
- They have the property of filling the entire part of a container by taking the shape of the container.
So, gases do not have fixed shape.
APPEARS IN
संबंधित प्रश्न
Give reasons:
A gas exerts pressure on the walls of the container.
Give reasons:
We can easily move our hand in air, but to do the same through a solid block of wood, we need a karate expert.
Which of the two diffuses faster : a liquid or a gas?
Fill in the following blank with suitable words :
Solid, liquid and gas are the three ........................ of matter.
State two characteristic properties of a gas.
Give two reasons to justify that Water is a liquid at room temperature.
A girl is cooking some food in the kitchen. The smell of food being cooked soon reaches her brother's room. Explain how the smell could have reached her brother's room.
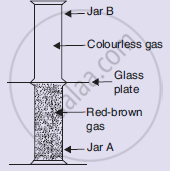
Look at the diagram on the right side. Jar A contains a red-brown gas whereas jar B contains a colourless gas. The two gas jars are separated by a galas plate placed between them
- What will happen when the glass plate between the two jars is pulled away?
- What name is given to the phenomenon which takes place?
- Name the brown gas which could be in jar A.
- Which is the colourless gas most likely to be present in jar B?
- Name one coloured solid and one colourless liquid which can show the same phenomenon.
A student placed a gas jar containing air in the upside down position over a gas jar full of red-brown bromine vapours. He observed that the red-brown colour spread upwards into the jar containing air. Based on this observation, the student concluded that it is only the bromine vapour which moves up and diffuses into air in the upper jar, the air from the upper jar does not move down by diffusion into the lower jar containing bromine vapours. Do you agree with this conclusion of the student? Give reason for your answer.
The space between ______ particles is the greatest one.
