Advertisements
Advertisements
Question
Why do gases not have fixed shape?
Solution
- Gases possess more intermolecular space and less forces of attraction.
- They can move freely at any direction.
- They have the property of filling the entire part of a container by taking the shape of the container.
So, gases do not have fixed shape.
APPEARS IN
RELATED QUESTIONS
Give reasons:
We can easily move our hand in air, but to do the same through a solid block of wood, we need a karate expert.
Which of the following diffuses fastest and which the slowest?
Solid, Liquid, Gas Give reasons for your answer.
Give two reasons to justify that Water is a liquid at room temperature.
When an incense stick (agarbatti) is lighted in one corner of a room, its fragrance quickly spreads in the entire room. Name the process involved in this.
When a gas jar full of air is placed upside down on a gas jar full of bromine vapours, the red-brown vapours of bromine from the lower jar go upward into the jar containing air. In this experiment :
Comment upon the following:-
rigidity
Comment upon the following:-
compressibility
Comment upon the following:-
density
Which of the following is false for gases?
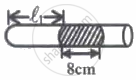 |
|
| Fig. I | Fig. II |
In figure-I, an air column of length ℓ1 is entrapped by a column of Hg of length 8 cm. In figure-II length of same air column at the same temperature is ℓ2. The `ℓ_1/ℓ_2` is:
(1 atm = 76 cm of Hg)