Advertisements
Advertisements
प्रश्न
Give reasons:
A gas exerts pressure on the walls of the container.
उत्तर
- Gas particles move randomly in all directions at high speed.
- As a result, the particles hit each other and also forcefully hit the walls of the container, exerting pressure on them.
APPEARS IN
संबंधित प्रश्न
Give a reason for the following observation.
We can get the smell of perfume sitting several metres away.
Fill in the following blank with suitable words :
Solid, liquid and gas are the three ........................ of matter.
Why do gases diffuse very fast?
Name two gases of air which dissolve in water by diffusion. What is the importance of this process in nature?
Comment upon the following:-
rigidity
Comment upon the following:-
shape
Comment upon the following:-
kinetic energy
Which of the following is false for gases?
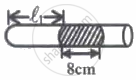 |
|
| Fig. I | Fig. II |
In figure-I, an air column of length ℓ1 is entrapped by a column of Hg of length 8 cm. In figure-II length of same air column at the same temperature is ℓ2. The `ℓ_1/ℓ_2` is:
(1 atm = 76 cm of Hg)
2 litre He gas at 2 atm and 300 K is inserted into a 4 litre rigid container containing N2 at 600 K and 4 atm. Finally mixture is maintained at 600 K temperature. The final pressure of gaseous mixture would be ______ torr.