Advertisements
Advertisements
प्रश्न
Why do gases diffuse very fast?
उत्तर
Gases diffuse very fast because of the presence of large spaces between the gaseous particles and high kinetic energy of these molecules. They diffuse faster than the other states of matter.
APPEARS IN
संबंधित प्रश्न
Comment upon the following:
rigidity, compressibility, fluidity, filling a gas container, shape, kinetic energy and density.
Fill in the following blank with suitable words :
Solid, liquid and gas are the three ........................ of matter.
Fill in the following blank with suitable words :
At room temperature, the forces of attraction between the particles of solid substances are ....................... then those which exist in the gaseous state.
Give two reasons to justify that Water is a liquid at room temperature.
A girl is cooking some food in the kitchen. The smell of food being cooked soon reaches her brother's room. Explain how the smell could have reached her brother's room.
Give reason for the following observation :
The smell of hot sizzling food reaches us even from a considerable distance but to get the smell from cold food, we have to go close to it.
Explain why, diffusion occurs more quickly in a gas than in a liquid.
Why does a gas exert pressure?
A substance has neither a fixed shape nor a fixed volume. State whether it is a solid, a liquid or a gas.
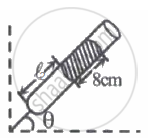
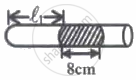 |
|
| Fig. I | Fig. II |
In figure-I, an air column of length ℓ1 is entrapped by a column of Hg of length 8 cm. In figure-II length of same air column at the same temperature is ℓ2. The `ℓ_1/ℓ_2` is:
(1 atm = 76 cm of Hg)