Advertisements
Advertisements
प्रश्न
Give reason for the following observation :
The smell of hot sizzling food reaches us even from a considerable distance but to get the smell from cold food, we have to go close to it.
उत्तर
The aroma of hot, sizzling food reaches us very easily even when we are far away from it, but the aroma of cold food reaches us only when we are near it because the rate of diffusion of a hot gas is faster than cold gas. When the gas is hot, the particles have greater kinetic energy and they move faster; therefore, they have a higher rate of diffusion.
APPEARS IN
संबंधित प्रश्न
The mass per unit volume of a substance is called density.
(density = mass/volume).
Arrange the following in order of increasing density-
air, exhaust from chimneys, honey, water, chalk, cotton and iron.
Name the physical state of matter which can be easily compressed.
Write the full forms of the following
- LPG
- CNG
If the fish is being fried in a neighbouring home, we can smell it sitting in our own home. Name the process which brings this smell to us.
Fill in the following blank with suitable words :
At room temperature, the forces of attraction between the particles of solid substances are ....................... then those which exist in the gaseous state.
Why does a gas exert pressure?
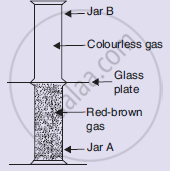
Look at the diagram on the right side. Jar A contains a red-brown gas whereas jar B contains a colourless gas. The two gas jars are separated by a galas plate placed between them
- What will happen when the glass plate between the two jars is pulled away?
- What name is given to the phenomenon which takes place?
- Name the brown gas which could be in jar A.
- Which is the colourless gas most likely to be present in jar B?
- Name one coloured solid and one colourless liquid which can show the same phenomenon.
Comment upon the following:-
filling a gas container
Explain why?
- A gas fills a vessel completely?
- Camphor disappears without leaving any residue.
2 litre He gas at 2 atm and 300 K is inserted into a 4 litre rigid container containing N2 at 600 K and 4 atm. Finally mixture is maintained at 600 K temperature. The final pressure of gaseous mixture would be ______ torr.
