Advertisements
Advertisements
प्रश्न
Fill in the following blank with suitable words :
At room temperature, the forces of attraction between the particles of solid substances are ....................... then those which exist in the gaseous state.
उत्तर
At room temperature, the forces of attraction between the articles of solid substances are much more than those which exist in the gaseous state.
APPEARS IN
संबंधित प्रश्न
Give reasons:
A gas exerts pressure on the walls of the container.
Write the full forms of the following
- LPG
- CNG
If the fish is being fried in a neighbouring home, we can smell it sitting in our own home. Name the process which brings this smell to us.
Name the three states of matter. Give one example of each.
State two characteristic properties of a gas.
When an incense stick (agarbatti) is lighted in one corner of a room, its fragrance quickly spreads in the entire room. Name the process involved in this.
Give one example of diffusion of gases in a liquid.
Why do gases diffuse very fast?
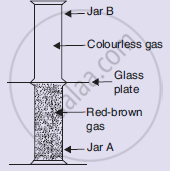
Look at the diagram on the right side. Jar A contains a red-brown gas whereas jar B contains a colourless gas. The two gas jars are separated by a galas plate placed between them
- What will happen when the glass plate between the two jars is pulled away?
- What name is given to the phenomenon which takes place?
- Name the brown gas which could be in jar A.
- Which is the colourless gas most likely to be present in jar B?
- Name one coloured solid and one colourless liquid which can show the same phenomenon.
Comment upon the following:-
density
