Advertisements
Advertisements
प्रश्न
Fill in the following blank with suitable words :
At room temperature, the forces of attraction between the particles of solid substances are ....................... then those which exist in the gaseous state.
उत्तर
At room temperature, the forces of attraction between the articles of solid substances are much more than those which exist in the gaseous state.
APPEARS IN
संबंधित प्रश्न
Tabulate the differences in the characteristics of states of matter.
Give reasons:
A gas exerts pressure on the walls of the container.
Write the full forms of the following
- LPG
- CNG
Which of the two diffuses faster : a liquid or a gas?
Explain how, the smell of food being cooked in the kitchen reaches us even from a considerable distance.
Why does a gas fill a vessel completely?
When a gas jar full of air is placed upside down on a gas jar full of bromine vapours, the red-brown vapours of bromine from the lower jar go upward into the jar containing air. In this experiment :
______ is not affected by gravity.
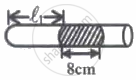 |
|
| Fig. I | Fig. II |
In figure-I, an air column of length ℓ1 is entrapped by a column of Hg of length 8 cm. In figure-II length of same air column at the same temperature is ℓ2. The `ℓ_1/ℓ_2` is:
(1 atm = 76 cm of Hg)
Two gases A and B having molecular weights 60 and 45 respectively are enclosed in a vessel. The wt. of A is 0.50 g and that of B is 0.2 g. The total pressure of the mixture is 750 mm. The partial pressure of the gases B is ______ mm Hg.