Advertisements
Advertisements
प्रश्न
Why does a gas fill a vessel completely?
उत्तर
Gases are easily compressible as they have large inter-molecular spaces. The gas particles can come closer to each other when external pressure is applied on gases.
On the other hand, it is almost impossible to compress a liquid because the intermolecular spaces in liquids are small. The particles in a solid are very close to each other. So, they cannot be compressed by applying external pressure.
APPEARS IN
संबंधित प्रश्न
Comment upon the following:
rigidity, compressibility, fluidity, filling a gas container, shape, kinetic energy and density.
Name the physical state of matter which can be easily compressed.
Fill in the following blank with suitable words :
Solid, liquid and gas are the three ........................ of matter.
Fill in the following blank with suitable words :
At room temperature, the forces of attraction between the particles of solid substances are ....................... then those which exist in the gaseous state.
State two characteristic properties of a gas.
When an incense stick (agarbatti) is lighted in one corner of a room, its fragrance quickly spreads in the entire room. Name the process involved in this.
What does the diffusion of gases tell us about their particles?
Explain why, diffusion occurs more quickly in a gas than in a liquid.
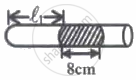 |
|
| Fig. I | Fig. II |
In figure-I, an air column of length ℓ1 is entrapped by a column of Hg of length 8 cm. In figure-II length of same air column at the same temperature is ℓ2. The `ℓ_1/ℓ_2` is:
(1 atm = 76 cm of Hg)
Two gases A and B having molecular weights 60 and 45 respectively are enclosed in a vessel. The wt. of A is 0.50 g and that of B is 0.2 g. The total pressure of the mixture is 750 mm. The partial pressure of the gases B is ______ mm Hg.