Advertisements
Advertisements
प्रश्न
Name the physical state of matter which can be easily compressed.
उत्तर
Gases can be compressed easily into small volumes because there are large spaces between their particles.
APPEARS IN
संबंधित प्रश्न
The mass per unit volume of a substance is called density.
(density = mass/volume).
Arrange the following in order of increasing density-
air, exhaust from chimneys, honey, water, chalk, cotton and iron.
If the fish is being fried in a neighbouring home, we can smell it sitting in our own home. Name the process which brings this smell to us.
Fill in the following blank with suitable words :
Solid, liquid and gas are the three ........................ of matter.
A girl is cooking some food in the kitchen. The smell of food being cooked soon reaches her brother's room. Explain how the smell could have reached her brother's room.
Explain how, the smell of food being cooked in the kitchen reaches us even from a considerable distance.
Why do gases diffuse very fast?
A student placed a gas jar containing air in the upside down position over a gas jar full of red-brown bromine vapours. He observed that the red-brown colour spread upwards into the jar containing air. Based on this observation, the student concluded that it is only the bromine vapour which moves up and diffuses into air in the upper jar, the air from the upper jar does not move down by diffusion into the lower jar containing bromine vapours. Do you agree with this conclusion of the student? Give reason for your answer.
The substance X normally exists in a physical state which can flow easily but does not fill its vessel completely. It also turns anhydrous copper sulphate blue. When substance X is cooled excessively, it changes into a substance Y which has a fixed shape as well as a fixed volume. If, however, the substance X is heated strongly, it changes into a substance Z which has neither a fixed shape nor a fixed volume.
- Name the substances (i) X (ii) Y and (iii) Z.
- What is the process of conversion of X into Y known as ?
- At which temperature X gets converted into Y ?
- What is the process of conversion of X into Z known as ?
- At which temperature X gets converted into Z ?
How does the substance of gaseous particles change to a liquid state?
 |
|
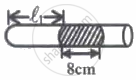
| Fig. I | Fig. II |
In figure-I, an air column of length ℓ1 is entrapped by a column of Hg of length 8 cm. In figure-II length of same air column at the same temperature is ℓ2. The `ℓ_1/ℓ_2` is:
(1 atm = 76 cm of Hg)