Advertisements
Advertisements
प्रश्न
A first order reaction is 50% completed in 1.26 × 1014 s. How much time would it take for 100% completion?
विकल्प
1.26 × 1015 s
2.52 × 1014 s
2.52 × 1028 s
infinite
उत्तर
infinite
Explanation:
Reaction would be 100% complete only after infinite time which cannot be calculated.
APPEARS IN
संबंधित प्रश्न
Time required to decompose SO2Cl2 to half of its initial amount is 60 minutes. If the decomposition is a first order reaction, calculate the rate constant of the reaction.
The rate constant for a first order reaction is 60 s−1. How much time will it take to reduce the initial concentration of the reactant to its `1/16`th value?
Straight line graph for first order reaction is obtained between ____________.
With the help of an example explain what is meant by pseudo first order reaction.
Observe the graph shown in figure and answer the following questions:
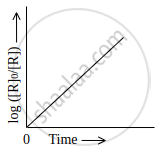
- What is the order of the reaction?
- What is the slope of the curve?
- Write the relationship between k and t1/2 (half life period).
Radioactive decay follows first-order kinetics. The initial amount of two radioactive elements X and Y is 1 gm each. What will be the ratio of X and Y after two days if their half-lives are 12 hours and 16 hours respectively?
What is the rate constant?
The following data were obtained during the decomposition of SO2Cl2 at the constant volume. SO2Cl2 →SO2(g) + Cl2(g)
| Time (s) | Total Pressure (bar) |
| 0 | 0.5 |
| 100 | 0.6 |
Calculate the rate constant of the reaction.
Write the equation for integrated rate law for a first order reaction.
Write the unit of rate constant [k] for the first order reaction.
