Advertisements
Advertisements
Question
A first order reaction is 50% completed in 1.26 × 1014 s. How much time would it take for 100% completion?
Options
1.26 × 1015 s
2.52 × 1014 s
2.52 × 1028 s
infinite
Solution
infinite
Explanation:
Reaction would be 100% complete only after infinite time which cannot be calculated.
APPEARS IN
RELATED QUESTIONS
The following data were obtained during the first order thermal decomposition of SO2Cl2 at a constant volume.
\[\ce{SO2Cl2_{(g)} -> SO2_{(g)} + Cl2_{(g)}}\]
| Experiment | Time/s–1 | Total pressure/atm |
| 1 | 0 | 0.5 |
| 2 | 100 | 0.6 |
Calculate the rate of the reaction when total pressure is 0.65 atm.
Which of the following graphs is correct for a first order reaction?

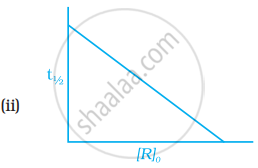
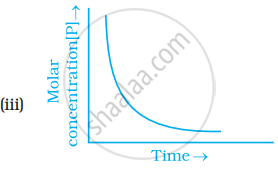
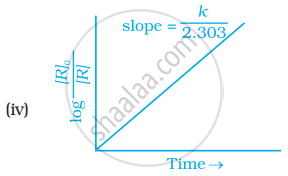
First order reaction is 50% complete in 1.26 × 1014s. How much time could it take for 100% completion?
A first order reaction is 50% complete in 20 minute What is rate constant?
In the first order reaction, half of the reaction is complete in 100 seconds. The time for 99% of the reaction to occurs will be
For a first order reaction, the ratio of the time for 75% completion of a reaction to the time for 50% completion is ______. (Integer answer)
The slope in the plot of ln[R] vs. time for a first order reaction is ______.
Radioactive decay follows first-order kinetics. The initial amount of two radioactive elements X and Y is 1 gm each. What will be the ratio of X and Y after two days if their half-lives are 12 hours and 16 hours respectively?
What is the rate constant?
Show that `t_(1/2)= 0.693/k` for first reaction.
