Advertisements
Advertisements
Question
Radioactive decay follows first-order kinetics. The initial amount of two radioactive elements X and Y is 1 gm each. What will be the ratio of X and Y after two days if their half-lives are 12 hours and 16 hours respectively?
Solution
For first-order reaction
half life of X = 12 hours
2 days = 48 hours means 4 half lives,
Amount of X left = `1/16` of initial value
half life of Y = 16 hours
2 days = 48 hours means 3 half lives,
Amount left = `1/8` of initial value
Ratio of X : Y = 1 : 2
APPEARS IN
RELATED QUESTIONS
Sucrose decomposes in acid solution into glucose and fructose according to the first order rate law with `"t"_(1/2)`= 3 hours. What fraction of the sample of sucrose remains after 8 hours?
Following data are obtained for reaction :
N2O5 → 2NO2 + 1/2O2
| t/s | 0 | 300 | 600 |
| [N2O5]/mol L–1 | 1.6 × 10-2 | 0.8 × 10–2 | 0.4 × 10–2 |
1) Show that it follows first order reaction.
2) Calculate the half-life.
(Given log 2 = 0.3010, log 4 = 0.6021)
A first order reaction takes 20 minutes for 25% decomposition. Calculate the time when 75% of the reaction will be completed.
(Given : log = 2 = 0·3010, log 3 = 0·4771, log 4 = 0·6021)
Which of the following graphs is correct for a first order reaction?

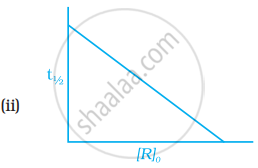
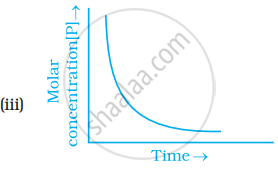
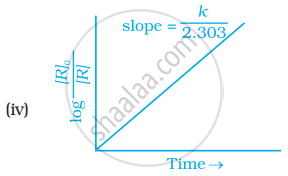
With the help of an example explain what is meant by pseudo first order reaction.
In the first order reaction, half of the reaction is complete in 100 seconds. The time for 99% of the reaction to occurs will be
In a first order reaction the concentration of reactants decreases from 400mol L-1 to 25 mol L-1 in 200 seconds. The rate constant for the reaction is ______.
The decomposition of formic acid on gold surface follows first-order kinetics. If the rate constant at 300 K is 1.0 × 10−3 s−1 and the activation energy Ea = 11.488 kJ mol−1, the rate constant at 200 K is ______ × 10−5 s−1. (Round off to the Nearest Integer)
(Given R = 8.314 J mol−1 K−1)
The reaction \[\ce{SO2Cl2(g) -> SO2(g) + Cl2(g)}\] is a first-order gas reaction with k = 2.2 × 10−5 sec−1 at 320°C. The percentage of SO2Cl2 is decomposed on heating this gas for 90 min, is ______%.
Write the unit of rate constant [k] for the first order reaction.
