Advertisements
Advertisements
प्रश्न
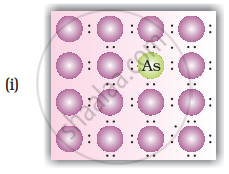
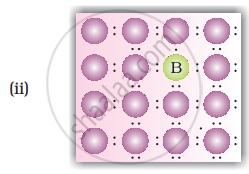
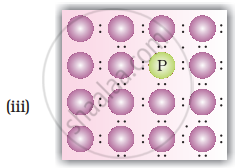
A perfect crystal of silicon (Figure) is doped with some elements as given in the options. Which of these options show n-type semiconductors?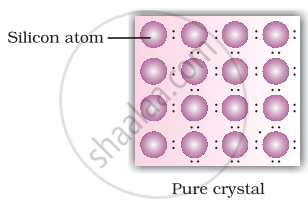
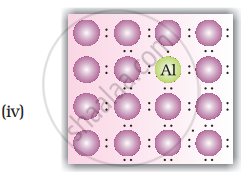
उत्तर


Explanation:
When group 15 elements are doped into a perfect crystal, it leads to the formation of n-type semiconductor. Here, in as (group 15, period 3) is doped to perfect Si-crystal and in as (group 15, period 2) is doped to perfect Si-crystal.
APPEARS IN
संबंधित प्रश्न
To prepare n-type semiconductor the impurity to be added to silicon should have the following number of valence electrons:
(a) 2
(b) 3
(c) 4
(d) 5
When a hole is produced in P-type semiconductor, there is _______.
A pure semiconductor is ________.
(a) an extrinsic semiconductor
(b) an intrinsic semiconductor
(c) p-type semiconductor
(d) n-type semiconductor
Group 14 element is converted to p-type semiconductor by doping it with:
In n-type semiconductor, current is carried by ____________.
Which transition metal oxide has appearance and conductivity like that of copper?
Which of the following oxides behaves as conductor or insulator depending upon temperature?
To get a n-type semiconductor from silicon, it should be doped with a substance with valence ______.
Explain why does conductivity of germanium crystals increase on doping with galium.
How does the doping increase the conductivity of semiconductors?
