Advertisements
Advertisements
Question
A perfect crystal of silicon (Figure) is doped with some elements as given in the options. Which of these options show n-type semiconductors?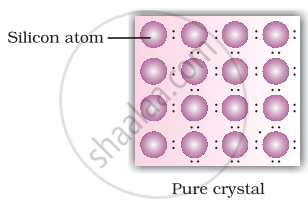
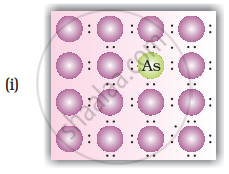
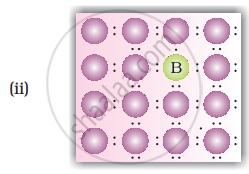
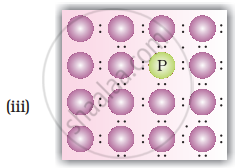
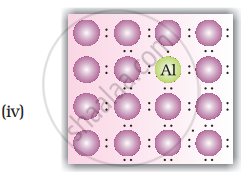
Solution


Explanation:
When group 15 elements are doped into a perfect crystal, it leads to the formation of n-type semiconductor. Here, in as (group 15, period 3) is doped to perfect Si-crystal and in as (group 15, period 2) is doped to perfect Si-crystal.
APPEARS IN
RELATED QUESTIONS
Distinguish between p-type and n-type semiconductors.
What is the ratio of octahedral holes to the number of anions in hexagonal closed packed structure?
A group of 14 elements is converted into n-type semiconductor by doping it with:
Group 14 element is converted to p-type semiconductor by doping it with:
Which transition metal oxide has appearance and conductivity like that of copper?
To get n-type of semiconductor, germanium should be doped with ____________.
Which of the following oxides behaves as conductor or insulator depending upon temperature?
To get a n-type semiconductor from silicon, it should be doped with a substance with valence ______.
Under the influence of electric field, which of the following statements is true about the movement of electrons and holes in a p-type semiconductor?
(i) Electron will move towards the positvely charged plate through electron holes.
(ii) Holes will appear to be moving towards the negatively charged plate.
(iii) Both electrons and holes appear to move towards the positively charged plate.
(iv) Movement of electrons is not related to the movement of holes.
How does the doping increase the conductivity of semiconductors?
